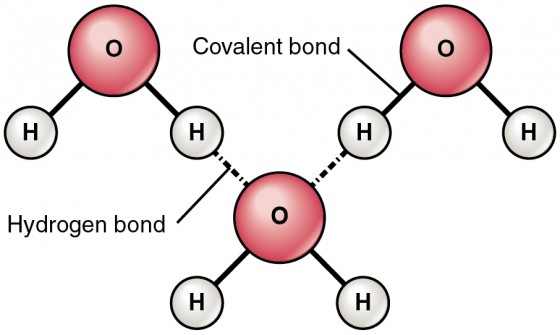
Hydrogen Bond in Water Molecule
Hydrogen Bonding of Water
|
The key to understanding water’s chemical behavior is its molecular structure. A water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent, forming a V shape. This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. All of the electron pairs (shared and unshared) repel each other, so the most stable arrangement is the one that puts them furthest apart from each other: a tetrahedron, with the O-H bonds forming two out of the four “legs.” The lone pairs are slightly more repulsive than the bond electrons, so the angle between the O-H bonds is slightly less than the 109° of a perfect tetrahedron, around 104.5°.
|
While there is no net charge on a water molecule, electrons are shared unequally between the oxygen atom and its hydrogen bond partners. Specifically, oxygen is more electronegative (electron-greedy) than hydrogen, so the shared electrons of the O-H bonds spend more of their time close to the oxygen than to the hydrogens. Since the electrons spend more time with the oxygen, the oxygen has a partial negative charge, while the hydrogens have partial positive charges. These polar covalent bonds, together with the bent geometry of molecule, make water apolar molecule.
|
Because each water molecule has positively and negatively charged regions, water molecules readily attract each other, with the plus end of one (a hydrogen atom) sticking to the minus end of another (an oxygen atom). These attractions are called hydrogen bonds. By definition, a hydrogen bond is a weak interaction between a hydrogen with a partial positive charge and a more electronegative atom (such as O). The hydrogens that participate in hydrogen bonding are attached to electronegative atoms, such as O, N, and F. Water molecules are also attracted to other polar molecules, as well as ions with full positive or negative charges. A charged or polar substance that interacts with and dissolves in water is said to be hydrophilic (hydro- = “water”; -philic = “loving”). In contrast, non-polar molecules such as oils and fats do not interact well with water, separate from it rather than dissolve in it, and are called hydrophobic (hydro- = “water”; -phobic = “fearing”). You may have noticed this as an inconvenient feature of salad dressings made of oil and vinegar. (Vinegar, in chemical terms, is just water with a bit of acid.)
|
References:
- Khan Academy. (n.d.). Retrieved December 29, 2015, from https://www.khanacademy.org/science/biology/water-acids-and-bases/hydrogen-bonding-in-water/a/hydrogen-bonding-in-water