Protein
Introduction
|
Proteins are among the most abundant organic molecules in living systems and are much more diverse in structure and function than other classes of macromolecules. A single cell in a living system can contain thousands of proteins, each with a unique function. Although their structures, like their functions, vary greatly, all proteins are made up of one or more chains of amino acids. In this article, we will look in more detail at the building blocks, structures, and roles of proteins.
|
Types and Functions of Protein
Proteins can play a wide array of roles in a cell or organism. Here, we’ll touch on a few examples of common protein types that may be familiar to you, and that are important in the biology of many organisms (including us).
|
Enzymes, which are produced by living cells, act as catalysts in biochemical reactions (meaning that they speed the reactions up). Each enzyme recognizes one or more substrates, the molecules that serve as starting material for the reaction it catalyzes. Different enzymes participate in different types of reactions and may break down, link up, or rearrange their substrates. One example of an enzyme found in your body is salivary amylase, which breaks amylose (a kind of starch) down into smaller sugars. The amylose doesn’t taste very sweet, but the smaller sugars do. This is why starchy foods often taste sweeter if you chew them for longer: you’re giving salivary amylase time to get to work.
|
Hormones are chemical signals released by endocrine cells (such as the cells of your pituitary gland) that control specific physiological processes, such as growth, development, metabolism, and reproduction. While some hormones are steroid-based (see the article on lipids), others are proteins. For example, insulin is an important protein hormone that helps regulate blood glucose levels.
|
Some additional types of proteins and their functions are listed in the table below:
Proteins come in many different shapes and sizes. Some are globular (roughly spherical) in shape, whereas others form long, thin fibers. For example, the hemoglobin protein that carries oxygen in the blood is a globular protein, while collagen, found in our skin, is a fibrous protein. A protein’s shape is critical to its function, and, as we’ll see in the next article, many different types of chemical bonds may be important in maintaining this shape. Changes in temperature and pH, as well as the presence of certain chemicals, may disrupt a protein’s shape and cause it to lose functionality, a process known as denaturation.
Amino Acids
|
Amino acids are the monomers that make up proteins, and all proteins are made of the same 20 amino acids. Well, okay, maybe that’s not 100% true. There are 20 canonical amino acids used to make proteins, but there are also three more unusual protein-building amino acids, used only by certain types of organisms or in response to certain signals in the genetic code (the instructions for producing a protein). We’ll look at these in more detail at the end of the section.
|
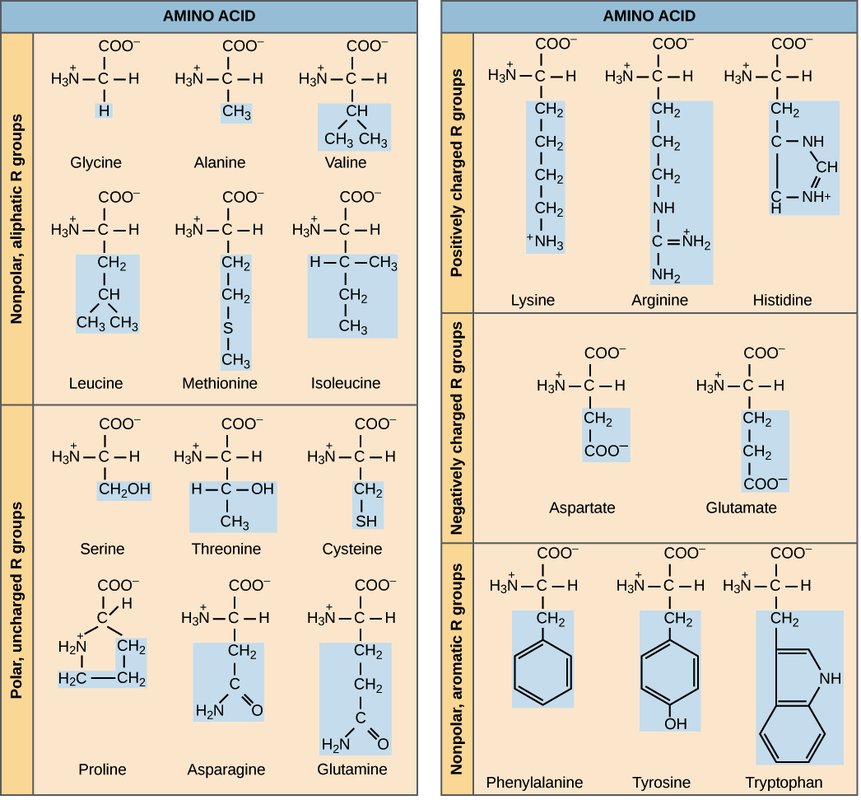
Each amino acid has the same basic structure, which consists of a central carbon atom, also known as the alpha (α) carbon, bonded to an amino group (NH_22start subscript, 2, end subscript), a carboxyl group (COOH), and a hydrogen atom. At physiological pH (7.2-7.4), the amino group is typically protonated and bears a positive charge, while the carboxyl group is typically deprotonated and bears a negative charge. Every amino acid also has another atom or group of atoms bonded to the central atom, known as the R group, which determines the identity of the amino acid. For instance, if the R group is a hydrogen atom, then the amino acid is glycine, while if it’s a methyl (CH_33start subscript, 3, end subscript) group, the amino acid is alanine. The twenty common amino acids are shown in the chart below, with their R groups highlighted in blue.
The properties of the side chain determine an amino acid’s chemical behavior (that is, whether it is considered acidic, basic, polar, or nonpolar). For example, amino acids such as valine and leucine are nonpolar and hydrophobic, while amino acids like serine and glutamine have hydrophilic side chains and are polar. Some amino acids, such as lysine and arginine, have side chains that are positively charged at physiological pH and are considered basic amino acids. (Histidine is sometimes put in this group too, although it is mostly deprotonated at physiological pH.) Aspartate and glutamate, on the other hand, are negatively charged at physiological pH and are considered acidic.
A few other amino acids have R groups with special properties, and these will prove to be important when we look at protein structure. For example, proline has an R group that’s linked back to its own amino group, forming a ring structure. Proline is thus an exception to the typical structure of an amino acid, since it no longer bears the standard NH_33start subscript, 3, end subscript^++start superscript, plus, end superscript amino group. If you think that ring structure looks a little awkward, you’re right: proline often causes bends or kinks in amino acid chains. Another special amino acid is cysteine, which contains a thiol (-SH) group and can form covalent bonds with other cysteines, which we’ll examine in more detail below.
Some of the twenty common amino acids cannot be synthesized by the body, but are necessary building blocks for some of the body’s proteins. These are known as essential amino acids, and in humans, they include isoleucine, leucine, and histidine. Since we cannot manufacture the essential amino acids, they must be absorbed from the food that we eat. The amino acids that are essential (must be obtained from external sources) varies from organism to organism.
A few other amino acids have R groups with special properties, and these will prove to be important when we look at protein structure. For example, proline has an R group that’s linked back to its own amino group, forming a ring structure. Proline is thus an exception to the typical structure of an amino acid, since it no longer bears the standard NH_33start subscript, 3, end subscript^++start superscript, plus, end superscript amino group. If you think that ring structure looks a little awkward, you’re right: proline often causes bends or kinks in amino acid chains. Another special amino acid is cysteine, which contains a thiol (-SH) group and can form covalent bonds with other cysteines, which we’ll examine in more detail below.
Some of the twenty common amino acids cannot be synthesized by the body, but are necessary building blocks for some of the body’s proteins. These are known as essential amino acids, and in humans, they include isoleucine, leucine, and histidine. Since we cannot manufacture the essential amino acids, they must be absorbed from the food that we eat. The amino acids that are essential (must be obtained from external sources) varies from organism to organism.
Peptide Bonds
|
The sequence and the number of amino acids in a protein ultimately determine that protein's shape, size, and function. Each amino acid is attached to its neighbors by a covalent bond known as a peptide bond, which is formed in a dehydration synthesis reaction. The carboxyl group of one amino acid and the amino group of the incoming amino acid combine, releasing a molecule of water. The resulting bond between amino acids is a peptide bond, and a long chain of amino acids linked by peptide bonds is known as a polypeptide. At one end, the polypeptide has a free amino group, and this end is called the amino terminus (or N-terminus). The other end, which has a free carboxyl group, is known as the carboxyl terminus (or C-terminus).
|
Protein Metabolism
|
During protein metabolism, some protein is converted to glucose in a process called gluconeogenesis, the formation of glucose from non-carbohydrate sources
The basic difference between protein and carbohydrate is that while carbohydrates are made out of simple sugars (carbon, hydrogen, and oxygen), protein is made from amino acids (carbon, hydrogen, oxygen, nitrogen, and sufur). The nitrogen is a basic component of the protein's amino acids and accounts for 13 to 20% of the total mass. The first step in protein metabolism is to break it into its constituent amino acids. These are absorbed into the blood stream. The second step is to break down the amino acids into their constituent parts--catabolism, if you want to get technical about it. This removes the nitrogen or amino group from the amino acids. The process is called deamination. |
Deamination breaks the amino group down into ammonia and what is termed the carbon skeleton. Ammonia is converted to urea, filtered through the kidneys, and excreted in urine. The carbon skeleton--which is composed of carbon, hydrogen, and oxygen--can then by used either for protein synthesis, energy production (ATP), or converted to glucose by gluconeogenesis.
Most authorities believe that the amount of protein converted to glucose is quite small, except under conditions of intense exercise or metablic starvation. Under these conditions amino acids produce the major source of glucose for blood sugar maintenance.
Most authorities believe that the amount of protein converted to glucose is quite small, except under conditions of intense exercise or metablic starvation. Under these conditions amino acids produce the major source of glucose for blood sugar maintenance.
|
In a person without diabetes, a rise in blood amino acid concentration (the result of protein metabolism) stimulates the secretion of both glucagon and insulin, so their blood sugar remains stable. But in people with diabetes, the release of glucagon without insulin or with impaired insulin response can cause our blood sugar to rise precipitously several hours after a meal high in protein.
The insulin is secreted to stimulate protein synthesis--the uptake of amino acids into muscle cells--making them less available for gluconeogenesis. The glucagon is secreted to stimulate the uptake of amino acids into the cells of the liver for gluconeogenesis. |
So why are these two hormones battling for opposing uses of the same amino acids? Isn't that non-productive?
Actually, the phenomenon serves an important purpose. As you probably know, insulin lowers the blood sugar, while glucagon raises it. In the non-diabetic state, the release of these two opposing hormones ensures that the amino acids are used for protein synthesis (because of the extra insulin) but the blood sugar doesn't drop to dangerously low levels, even if the meal was low in carbohydrate. As a result, blood glucose concentration remains reasonably stable during protein metabolism. The insulin and glucagon essentially cancel each other out in terms of their effect on blood glucose, while the insulin is still able to promote protein synthesis.
But in people with diabetes, as I mentioned earlier, the release of glucagon without insulin or with impaired insulin response can cause our blood sugar to rise precipitously several hours after a meal high in protein. This is due not only to the glucagon's directly raising the blood sugar, but also to the fact that in the absence of insulin it increases the amount of the amino acids that are used for gluconeogenesis.
Actually, the phenomenon serves an important purpose. As you probably know, insulin lowers the blood sugar, while glucagon raises it. In the non-diabetic state, the release of these two opposing hormones ensures that the amino acids are used for protein synthesis (because of the extra insulin) but the blood sugar doesn't drop to dangerously low levels, even if the meal was low in carbohydrate. As a result, blood glucose concentration remains reasonably stable during protein metabolism. The insulin and glucagon essentially cancel each other out in terms of their effect on blood glucose, while the insulin is still able to promote protein synthesis.
But in people with diabetes, as I mentioned earlier, the release of glucagon without insulin or with impaired insulin response can cause our blood sugar to rise precipitously several hours after a meal high in protein. This is due not only to the glucagon's directly raising the blood sugar, but also to the fact that in the absence of insulin it increases the amount of the amino acids that are used for gluconeogenesis.
References:
- Khan Academy. (n.d.). Retrieved December 18, 2015, from https://www.khanacademy.org/science/biology/macromolecules/proteins-and-amino-acids/a/introduction-to-proteins-and-amino-acids