ATP (Adenosine triphosphate)
|
Energy
When the third phosphate group of ATP is removed by hydrolysis, a substantial amount of free energy is released. The exact amount depends on the conditions, but we shall use a value of 7.3 kcal per mole.
ATP + H2O → ADP + Pi
When the third phosphate group of ATP is removed by hydrolysis, a substantial amount of free energy is released. The exact amount depends on the conditions, but we shall use a value of 7.3 kcal per mole.
ATP + H2O → ADP + Pi
ADP is adenosine diphosphate. Pi is inorganic phosphate.
Because of the substantial amount of energy liberated when it is broken, the bond between the second and third phosphates is commonly described as a "high-energy" bond and is depicted in the figure by a wavy red line. (The bond between the first and second phosphates is also "high-energy".) (But please note that the term is not being used in the same sense as the term "bond energy". In fact, these bonds are actually weak bonds with low bond energies.)
Cells contain a wide variety of enzymes — called ATPases — that catalyze the hydrolysis of ATP and couple the energy released to particular energy-consuming reactions in the cell
|
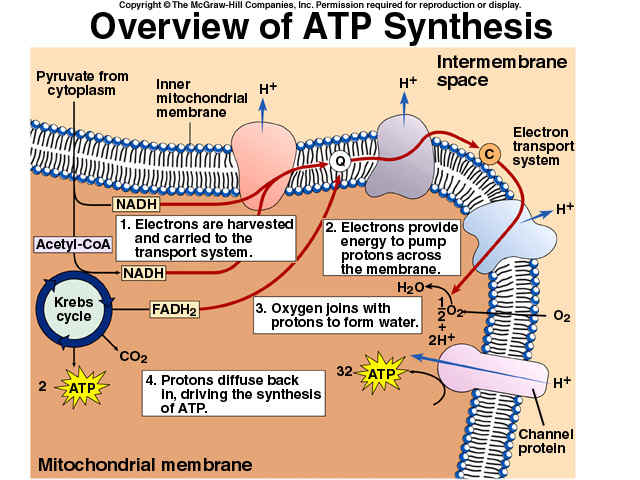
Synthesis of ATP
|
Consumption of ATP
ATP powers most of the energy-consuming activities of cells, such as:
|
Extracellular ATP
In mammals, ATP also functions outside of cells. Its release
In mammals, ATP also functions outside of cells. Its release
- from damaged cells can elicit inflammation and pain;
- from the carotid body signals a shortage of oxygen in the blood;
- from taste receptor cells triggers action potentials in the sensory nerves leading back to the brain;
- from the stretched wall of the urinary bladder signals when the bladder needs emptying.
References:
- ATP (Adenosine triphosphate). (n.d.). Retrieved December 22, 2015, from http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/A/ATP.html